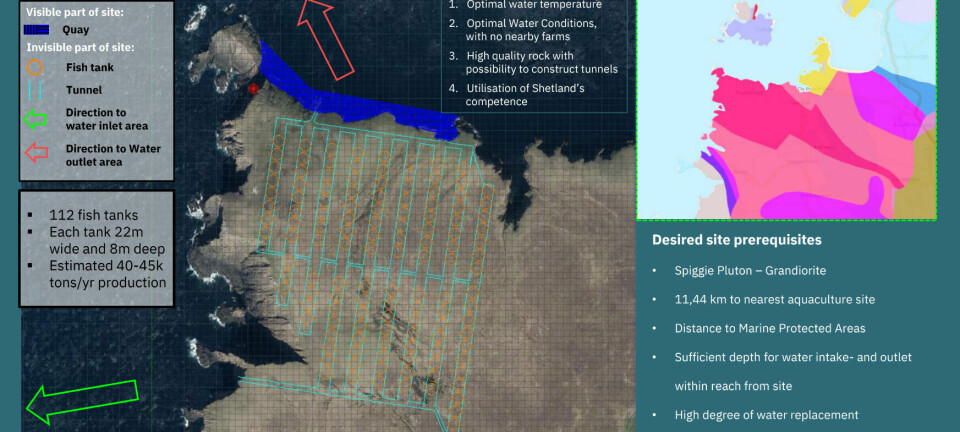
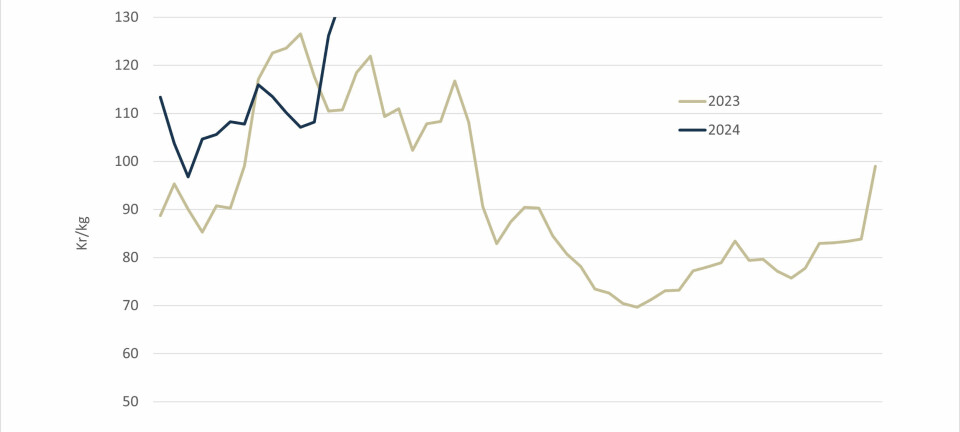
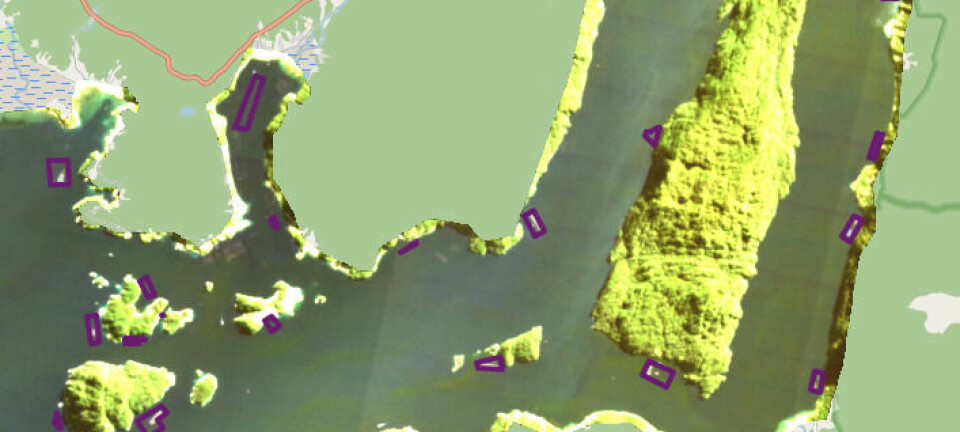
SLICE study may take two years to complete
Detailed research into the environmental effects of using in-feed sea louse treatment SLICE is likely to take up to two years, according to Scott Landsburgh, chief executive of the Scottish Salmon Producers’ Organisation (SSPO).
The SSPO will commission the research in response to the decision of the Scottish Environmental Protection Agency (SEPA) to cut back on the use of emamectin benzoate (SLICE) by 60 per cent.
And Landsburgh revealed that WRc, the firm which carried out the study on emamectin benzoate that prompted SEPA’s action, is likely to be one of the firms asked to tender for the SSPO work.
“We have still got to go through the commissioning process,” said Landsburgh. “One of the companies we will likely approach to pitch for the work will be WRc because they carried out the desk study for SEPA so we think we should at least give them the opportunity to pitch for this study as well.
“Our study would take physical evidence from the marine waters of Scotland, including benthic samples.

“It cannot be as quick as the timescale for the consultation period (which closes at the end of August). This research will take a couple of years. We are busy writing the scope of it just now and once we have completed that we will put it out for tender, but we do think that we would like WRc to be one of the firms to tender."
Comprehensive
“We anticipate this will take a couple of years because it will be comprehensive and also we need to assess over a significant period, it can’t just be done in a matter of weeks," said Landsburgh. "It will be absolutely relevant to the current and historical use of SLICE in our waters, which is how things should be measured and assessed as opposed to just looking at theoretical research based on desk studies.
“We are working on the scope of the study as of now and have been since the publication of the WRc report by SEPA and we hope to be in a position to give further details at the end of August by the latest.”
Referring to the fact that SEPA’s deadline for consultation on its proposals to reduce the use of SLICE is August 31, Landsburgh said: “We will obviously make some reference to the fact that we are commissioning further research.
“We will certainly be going back to SEPA now and asking them to take this into consideration and to seek an extension of the consultation period for a number of reasons. One is that they have put out this very short [consultation] period over the summer months when a lot of our scientists within the industry are on their holidays, and therefore it is difficult for us to garner sufficient scientific background knowledge for us to fulfil all the consultation response requests.
“We will certainly put in a consultation response which will be appropriate based upon what we know so far but equally we be looking for consideration of what will be the outcome of our report in due course.”
Mitigation measures
SEPA announced last week that it is putting in place an interim regulatory position for new applications that limits residues in sediment of emamectin benzoate “to the extent that practically useable quantities are unlikely to be able to be authorised, unless effective mitigation measures are put in place to collect fish faeces and ensure the metabolites from the administration of the medicated feed are contained”.
Operators at existing sites are being asked to reduce their application of emamectin benzoate by 60 per cent.
SEPA is concerned that emamectin benzoate in fish faeces and uneaten food might harm animals that live on the seabed under fish farms, such as crabs, which are related to the sea lice the medicine is designed to kill.
After SEPA’s announcement Landsburgh said: “SEPA have published a report based on desk research on non-native species in freshwater, regardless of the fact that our salmon mature in marine water. Then they ask if anyone has any other information to contribute to their findings. No business can operate successfully in this sort of chaos.”
He added: “SLICE has been, and continues to be, a critically important tool within the industry’s strategy to protect the health and welfare of farmed salmon.”